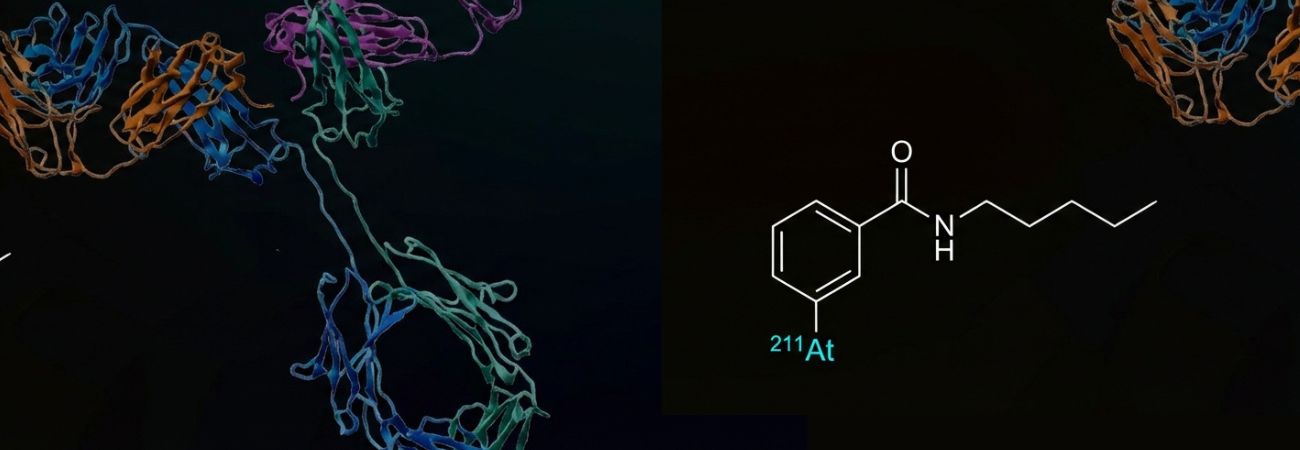
Technology
A disruptive innovation in the treatment of microscopic residual disease
Therapeutic innovation
The technology developed by Atonco is a highly promising disruptive innovation in the eradication of microscopic residual disease or isolated or clustered residual tumor cells that are rapidly accessible to a radiopharmaceutical labeled with astatine-211.
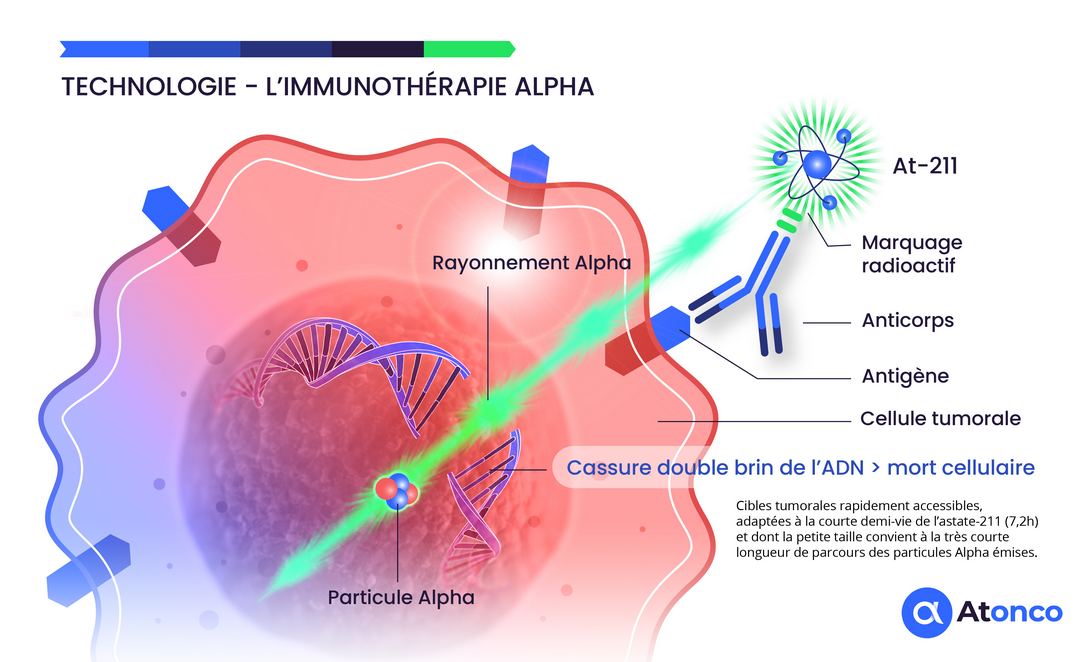
The use of alpha particle-emitting radionuclides is a current topic of great medical interest due to the high cytotoxic effect of these alpha particles on tumor cells, which results in double-strand DNA breaks. Furthermore, this approach offers good tolerability because the short range of alpha particles potentially spares normal tissues close to tumor targets.
Why Astatine-211?
Astatine-211 (²¹¹At) represents the optimal alpha-emitting radionuclide for targeted therapy due to its unique physical and chemical properties. Its half-life of 7.2 hours offers an ideal balance between the time required for production, purification, conjugation, and therapeutic administration, while minimizing prolonged systemic exposure.
The alpha particles emitted by ²¹¹At have an energy of 5.9 MeV with a tissue range of 55–80 μm, corresponding to 5–10 cell diameters. This characteristic enables selective destruction of targeted tumor cells while preserving adjacent healthy tissue, offering an exceptionally favorable therapeutic index.
The high linear energy transfer (LET) of ~100 keV/μm induces irreparable DNA damage, independent of the oxygenation status of tumor cells. This property is particularly advantageous for the treatment of hypoxic tumors, which are often resistant to conventional therapies.
Bibliography
Clinical application
Alpha-immunotherapy of non-muscle-invasive bladder cancer (NMIBC) with a chimeric anti-Carbonic Anhydrase IX (CAIX) antibody, girentuximab, labeled with astatine-211: our candidate, ATO-101™
ATO-101™ targets non-muscle-invasive bladder tumors that are refractory to BCG and chemotherapy and pending total cystectomy.
ATO-101™, our radiopharmaceutical candidate, is an anti-CAIX antibody labeled with astatine-211 using a proprietary radiolabeling technology developed in Nantes, France.
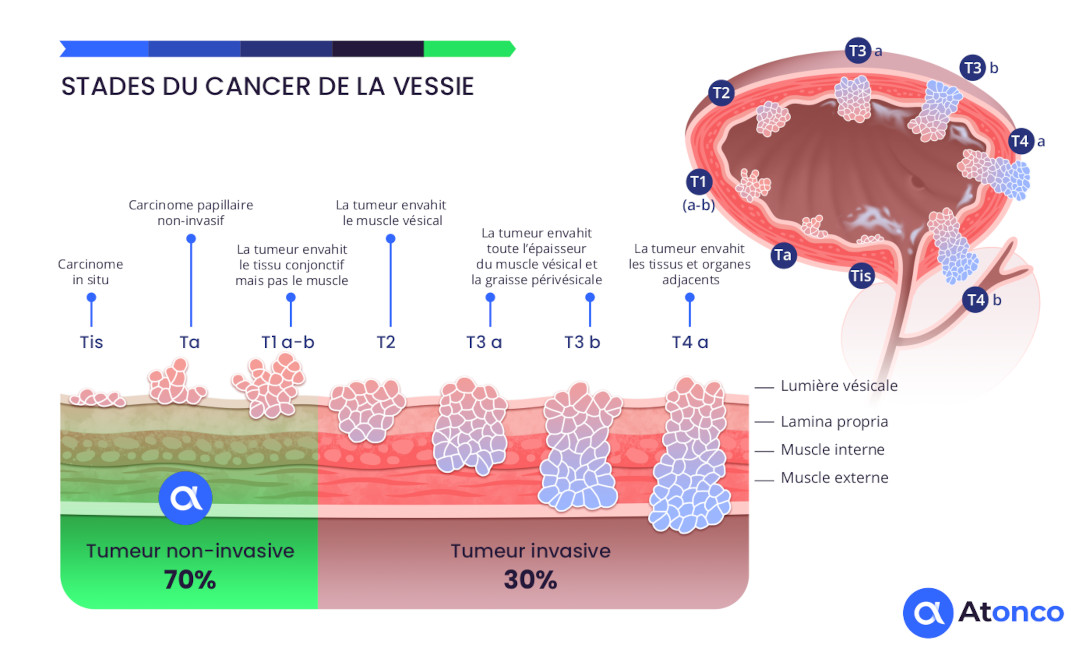
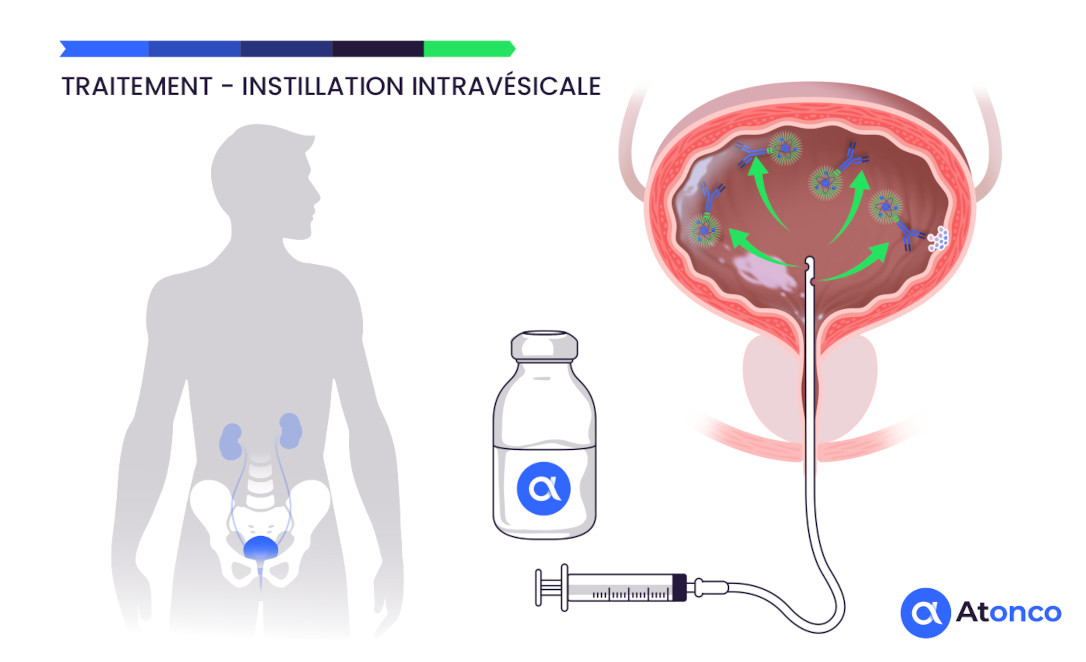
The CAIX antigen is expressed on the membrane of cancer cells of superficial non-muscle-invasive bladder tumors and is therefore directly and rapidly accessible to the radiolabeled anti-CAIX antibody instilled intravesically. Furthermore, recurrences that frequently occur after repeated BCG or chemotherapy treatments consist of thin layers of cancer cells that respond well to the very short path length of the emitted alpha particles. Thus, the non-muscle-invasive bladder tumor setting is well suited to the radiophysical characteristics of alpha particles.

The tumors have a thin thickness well suited to the very short path length (<0.1 mm) of the alpha particles, and their superficial location in the bladder cavity makes them accessible to a radiopharmaceutical instilled intravesically. Moreover, the clinical situation of patients refractory to conventional BCG therapy or chemotherapy, for whom the risk of an uncomfortable total cystectomy justifies the implementation of the innovative alpha-immunotherapy delivered by intravesical instillation.
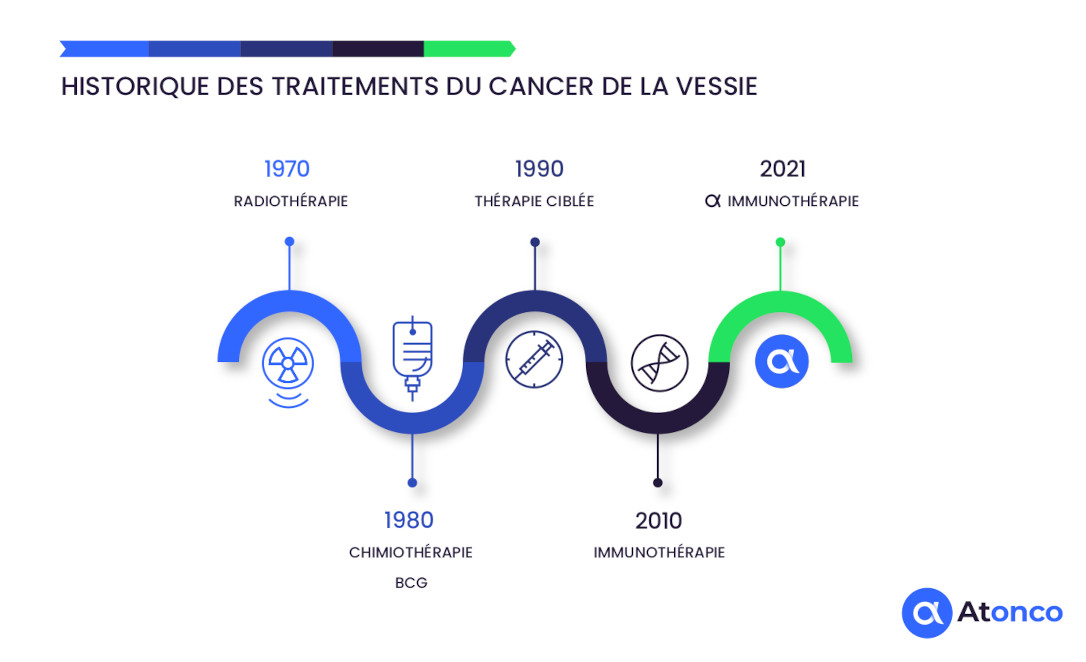
History of bladder cancer treatments
Stages of bladder cancer
Treatment – Intravesical instillation
Bibliography
Innovative treatment
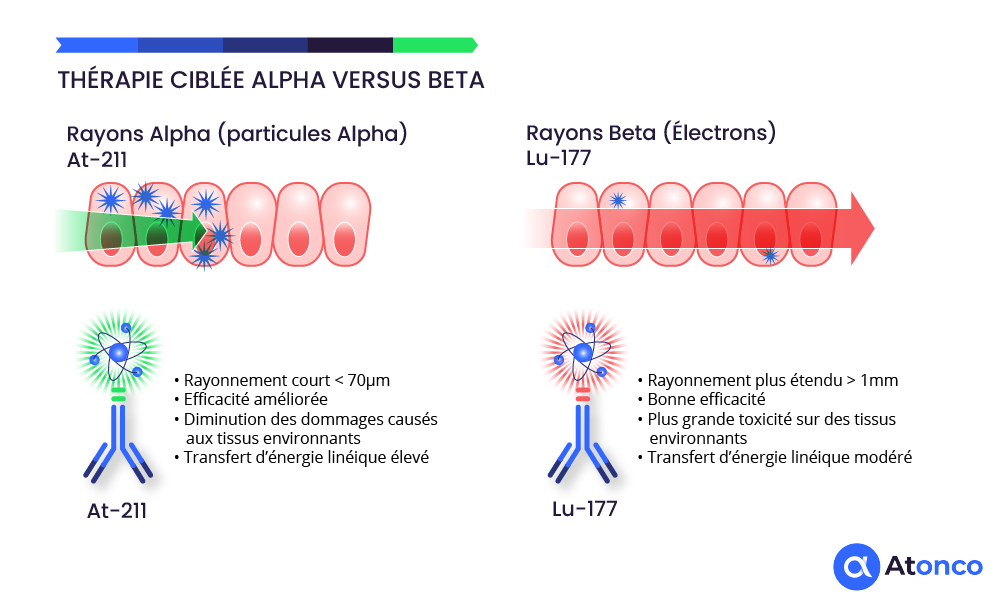
Targeted alpha therapy using Astatine-211 represents a major advance in the treatment of cancer, offering decisive advantages over conventional therapeutic approaches.
Advantages of alpha particles over conventional treatments
Alpha particles offer decisive advantages in killing tumor cells compared to external radiotherapy and chemotherapy, notably through their superior biological efficacy and limited toxicity to surrounding healthy tissues. Their very high linear energy transfer (100 times greater than that of beta radiation/electrons) induces irreparable double-strand DNA breaks, leading to irreversible destruction of cancer cells. Moreover, the very short range of alpha particles (only a few cell layers) concentrates the dose locally, reducing damage to nearby healthy cells and limiting the unwanted side effects often observed with external radiotherapy and chemotherapy.
Biological efficacy and mechanism of action
Unlike treatments such as chemotherapy that require prolonged cellular internalization, targeted alpha therapy can kill tumor cells through the immediate damage caused by alpha particles to DNA without requiring internalization of the vector. One to three alpha particles are sufficient to kill a tumor cell, whereas thousands of beta particles or chemotherapy molecules are required. This targeted efficacy is particularly well suited to micro-metastases and small residual tumor clusters, as in the case of non-muscle-invasive bladder cancer following resection of macroscopic lesions.
Limited toxicity and precise targeting
The short range of alpha particles in tissue (2 to 5 cell layers) allows a very local concentration of the dose, thereby reducing toxicity to surrounding healthy tissues, unlike more diffuse external radiotherapy or chemotherapy which can affect multiple cell types. This targeting is made possible through the use of biological vectors (antibodies, peptides) that carry the alpha radioisotope directly to tumor cells, reducing the area of exposure and systemic side effects.
Comparison with external radiotherapy and chemotherapy
External radiotherapy: often treats a broad tumor and surrounding area, thereby exposing healthy tissues to radiation and potentially causing significant side effects. Its wider penetration is less suited to small, dispersed lesions.
Chemotherapy: acts systemically, also affecting healthy cells with multiple toxicities, particularly hematological and gastrointestinal.
Alpha therapy: precisely targets cancer cells, minimizes collateral damage, and optimizes cell destruction even in difficult-to-reach areas such as micro-metastases.
These advantages make alpha particle therapy a promising and complementary therapeutic approach, particularly in cancers resistant to conventional treatments, offering a better efficacy-to-toxicity ratio.
